  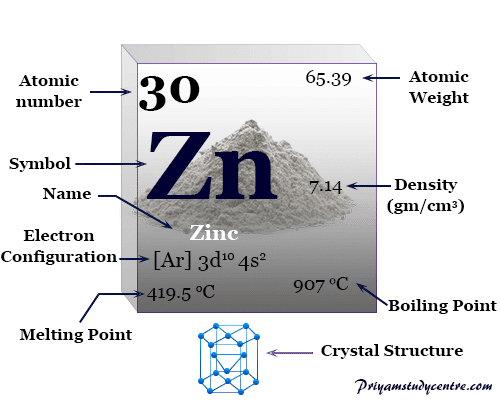
It is the first element in group 12 (IIB) of the periodic table. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. Zinc is a shiny solid at standard temperature and pressure and has a high melting point. Zinc is a chemical element it has symbol Zn and atomic number 30. Every hydrogen atom has one proton in its nucleus. Hydrogen, at the upper left of the table, has an atomic number of 1. In this table, an element's atomic number is indicated above the elemental symbol. Zinc is a metal element so it is a good thermal conductor and a good electrical conductor. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. Properties Zinc forms ionic bonds with non-metals. 
Zinc loses electrons to form positive metal ions. Zinc is in Period 4 of the Periodic Table because it has 4 electron shells. Atomic Structure The most stable isotope of Zinc has 35 neutrons in its nucleus giving it an atomic mass of 65. Zinc atoms join together in a giant metallic structure. However, even these small quantities are required for the body to function properly.Zinc is a transition metal element, on the Periodic Table, with 30 protons in the nucleus.Ībout Zinc Molecular Structure Zinc has the chemical formula Zn. These last three metals are present in the body in very small quantities. Cobalt is a necessary component of vitamin B-12, a vital nutrient. Manganese is needed for the body to metabolize oxygen properly. Copper is also needed for several proteins to function properly in the body. Zinc is needed for the body’s immune system to function properly, as well as for protein synthesis and tissue and cell growth. Other transition metals have important functions in the body, despite being present in low amounts. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. Zinc burns in air at high red heat, evolving white clouds of zinc. It is brittle at low temperatures but becomes malleable at 100-150C. The critical part of the red blood cell is a protein called hemoglobin. Zinc has a melting point of 419.58C, a boiling point of 907C, a specific gravity of 7.133 (25C), with a valence of 2. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. Iron is a transition metal and the chemistry of iron makes it a key component in the proper functioning of red blood cells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The most abundant non-main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. An elements mass is listed as the average of all its isotopes on earth. This mass is usually an average of the abundant forms of that element found on earth. Elements are either metals, nonmetals, or semimetals. The number of moles in a system can be determined using the atomic mass of an element, which can be found on the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
